1:30min

Professor Robyn Guymer Photo: CERA
______________________________
By Helen Carter
Journalist
Australian patients are needed to join three global clinical trials of world-first treatments for dry age-related macular degeneration.
Ophthalmologist Professor Robyn Guymer, head of Macular Research at the Centre for Eye Research Australia (CERA), is calling on Victorian optometrists and ophthalmologists to refer dry AMD patients to the ground-breaking trials, some of which are already underway.
CERA is involved in all three multi-centre, international, industry, treatment trials sponsored by pharmaceutical companies, Allergan, Roche and Apellis.
‘It’s the first time there has been a possible treatment for dry AMD. We’ve had treatments for neovascular or wet AMD for 10 years and these drugs have more than halved the blindness rate in wet AMD,’ Professor Guymer said.
‘For nearly 20 years I’ve seen patients with geographic atrophy (dry AMD) in the clinic and apart from monitoring their vision loss over the years, there was nothing we could do for them. Now we finally have the possibility of some treatment options on the horizon, and we need optometrists and eye-health providers to spread the word and refer people who potentially meet our criteria.’
Professor Guymer, who is lead investigator of the Melbourne trials, said the neuroprotective and anti-inflammatory drugs aimed to keep unhealthy neurons alive longer.
‘There are three trials, trialling different modes and treatment intervals,’ she said. ‘Dry AMD treatment is injected into the vitreous, similar to wet AMD treatment injections. Some patients will receive a fluid form and the others will have a slow-release device injected, which is long-acting. Others will be control patients.’
The trials will compare efficacy of injections every four versus six or eight weeks, and the slow-release form which is injected much less frequently, every three months.
The three trials will be conducted for two years. During this time, researchers will measure the success of the drug and determine its efficacy by measuring the size and rate of growth of atrophic holes in each patient’s macula and the number of atrophic holes. Autofluorescence photos will measure the surface area of these holes.
‘Dry AMD is like having moth-eaten holes in your vision. There is a slow growth of those holes,’ Professor Guymer told Australian Optometry.
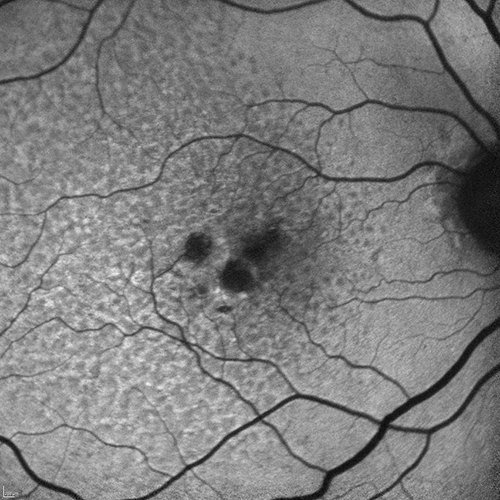
Autofluorescence images of areas of degenerative macula in geographic atrophy Image: Professor Robyn Guymer, CERA
She said phase one studies of the drugs were promising and all treatments had slowed the rate of growth of atrophic holes in the retina, so they had showed safety and some efficacy. These phase two and three studies aimed to further establish efficacy by determining if treatments could delay disease progression.
‘In geographic atrophy vision loss is very slow,’ Professor Guymer said.
‘With wet AMD people can lose vision within weeks after turning up at a hospital emergency department with a haemorrhage in their eye. Dry AMD is more insidious because people are not sure they have anything wrong and vision loss sneaks up until it’s almost too late.
‘Central vision can be good until the end yet reading can be very difficult due to the missing pieces of words or numbers. It’s slow because cells drop out little by little over a long period of time.’
About 57,000 Australians have dry AMD, threatening their vision, according to the Macular Disease Foundation Australia’s Macular Degeneration Facts and Figures report, 2012.
Professor Guymer said health professionals currently had no idea how to treat dry AMD other than to give general health advice.
Patients eligible for the studies can have dry AMD in one or both eyes.
‘People with just early disease (drusen or pigmentation change in both eyes) are not eligible. ‘Participants have to be at least end-stage geographic atrophy in one eye,’ Professor Guymer said. ‘These people might not know they have vision loss but might have difficulty reading and lose their place or misread numbers or miss bits of the scene that are not quite central.’
Eye health professionals can inform their dry AMD patients about the trials. If they wish to take part, they can send the patient’s details, retinal diagnosis and scans or images by email to CERA, or post them to Professor Guymer, CERA, Level 1, 32 Gisborne-Street, East Melbourne VIC 3002.
Their information will be saved to a registry of potential participants to ensure they can take part in future trials, if they are not eligible for current trials.
CERA requires from 10 to 20 people for each of the three trials. Those not eligible for the clinical treatment trials may be able to be enrolled in one of several natural history trials of dry AMD patients also underway, where no treatment intervention is given but patients are followed and have a six-monthly check-up.
Optometrists can also refer patients to the trials interstate.
Allergan is conducting its trial in Perth. Optometrists may refer patients directly to the principal investigator, Associate Professor Fred Chen, at the Lions Eye Institute, 2 Verdun Street, Nedlands WA 6009 or email fredchen@lei.org.au or fcreceptionist@lei.org.au.
Two studies sponsored by Roche to investigate treatments for AMD are recruiting patients in Australia. The Chroma (GX29176) study is being conducted in Sydney, Albury, Parramatta and Westmead, and the Spectri (GX29185) study is being conducted in Box Hill, Perth, Brisbane and Adelaide.
Optometrists who require additional information can contact Roche Medical Information at 1800 233 950 or email Australia.medinfo@roche.com
Read about the Appelis Pharmaceuticals treatment APL-2 (intravitreal).