1:30min

By Helen Carter
A new alternative to lifelong glaucoma eye-drops is available in Australia for cataract surgery patients with glaucoma.
Micro-invasive glaucoma surgery involves inserting a titanium stent smaller than a pinhead through a 1.5 mm corneal incision.
This creates a permanent opening in the trabecular meshwork to continuously improve the outflow of aqueous humour from the eyes, therefore lowering pressure within the eye.
Ophthalmologist Dr Brendan Cronin said the iStent was approved by the Therapeutic Goods Administration in January for use in patients with mild to moderate open-angle glaucoma undergoing cataract surgery.
It is the tiniest medical implant in the world and the smallest medical device approved by America’s Food and Drug Administration.
Dr Cronin is a corneal and anterior segment specialist and director of education at Queensland Eye Institute. He will explain the government-funded procedure to registrants at the Australian Vision Convention (AVC), which is being organised by Queensland and Northern Territory Division, on the Gold Coast in April.
 |
| Dr Brendan Cronin |
‘Optometrists can now advise glaucoma patients there is a surgical alternative to drops, provided they have had cataract surgery,’ Dr Cronin said.
‘These devices will become basic standard of care over the next couple of years. This will become mainstream glaucoma management for ophthalmologists.
‘Optometrists need to know about it as they will refer patients for the surgery and will see them afterwards to check intraocular pressure,’ he said.
‘It is very different from traditional glaucoma surgery for severe cases where eye-drops are not working.
‘This implant is suitable for patients with mild to moderate glaucoma and is great because it means they no longer have to be dependent on their drops. Evidence shows it is efficacious at keeping IOP down. It is a lifelong therapy as the stent will stay in forever.
‘Patients love it because there are no more eye-drops. People hate drops—they forget to take them, they are time-consuming and they can sting.
‘It is important patients realise this is not a cure for glaucoma. They will still have the disease and still need regular check-ups with their optometrist or ophthalmologist to check IOP.’
Dr Cronin said that some patients may still need to use eye-drops but the stent would be likely to reduce the amount required.
‘The iStent bypasses the pathway of resistance in glaucoma. It has minimal side-effects, is very simple to use and is a quick operation done while a patient is having cataract surgery,’ he said.
‘It can also be done years after they have had cataract surgery but only cataract patients can receive the implant as it is implanted in the incision for cataract surgery.’
Patients cannot see or feel the stent after the procedure.
Dr Cronin learned the surgical technique in the UK in 2012 and performed the procedure there. He said the implants had been done for several years in the UK and up to four years in Europe.
Following the TGA approval, he will now start performing the surgery here.
‘I think it’s going to explode in Australia. A handful of ophthalmologists have been doing this but soon there will be hundreds,’ he said.
‘There are more stents becoming available very soon and I am an investigator with Ivantis and its new hydros stent which is TGA approved but not available to anyone outside the investigational group yet.’
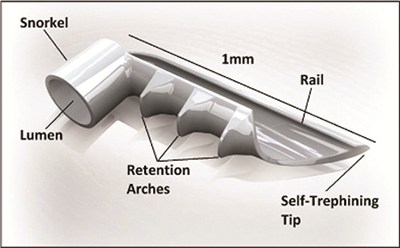
The iStent is manufactured by Glaukos and imported by Designs For Vision Australia. General manager Brock Flowers said it had been previously implanted in Australia in a pilot study.
‘The iStent is available now through a limited number of surgeons who have undergone the specific training on its use and insertion,’ Mr Flowers said.
‘It will be available to both the private and public systems and there is an item number for the surgical procedure under the Medicare Benefits Schedule.’
The Australian Register of Therapeutic Goods states: ‘The iStent Trabecular Micro Bypass Stent is indicated for use in conjunction with cataract surgery for the reduction of IOP in subjects with mild to moderate open angle glaucoma currently treated with ocular hypotensive medication.’
- Dr Cronin will be speaking at AVC on 26 April. The conference on 25-27 April will be at the Gold Coast Convention and Exhibition Centre and provides up to 51 CPD points, 20 of which are therapeutic. Register at www.etouches.com/avc2014.